by Kathleen Haughney, Florida State University
Create Blue Light from a Promising Material: A Florida State University research team has developed a new way to create blue light from a class of materials that shows enormous potential for optoelectronic devices, including solar cells, light-emitting diodes (LEDs) and lasers.
Led by FSU Professor of Chemistry Biwu Ma, the team has published a new study in Advanced Energy Materials that lays out a simple and effective approach to create an efficient and stable blue light from metal halide perovskites. Metal halide perovskites are an emerging class of semiconductor materials characterized by a cubic crystal structure that have good light-emitting properties while also being easy and inexpensive to manufacture.
Scientists, including Ma, have already created highly efficient and stable perovskite-based LEDs for green and red light, but an efficient and stable blue light has been difficult to achieve. Blue light requires a lot of power, and the blue color purity often decreases over time.
Without an efficient and stable blue light, it is impossible to create white light.
“We developed new strategies to achieve efficient and stable blue light from perovskites and fabricate LEDs with higher performance,” Ma said. “If you want a full color display, you need blue, green and red. Green and red have a good performance already, but blue is harder. It’s not easy to make a stable blue because it has higher energy.”
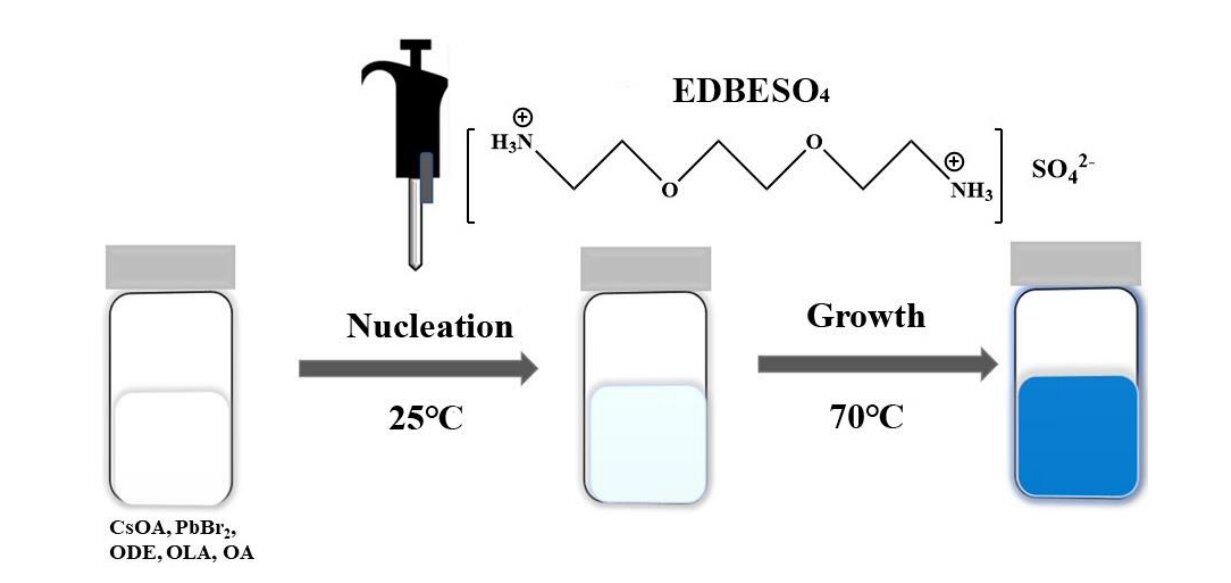
Ma and his team created blue emitting nanoplatelets using a metal halide perovskite based on the chemical compound cesium lead bromide, or CsPbBr3. Nanoplatelets are nanomaterials with only a few unit cells in thickness and, as a result, experience the effects of strong dielectric and quantum confinement.
To make these particular nanoplatelets emit efficient and stable blue light, researchers coated them with a multifunctional organic sulfate that allowed for what’s called surface passivation, a highly effective method used to improve luminescence properties and stability.
In this project, the simple organic sulfate passivation helps keep the nanoplatelets from degrading, allowing them to emit a more efficient and stable blue light.
With these surfaced passivated CsPbBr3 nanoplatelets as emitters, proof-of-concept LEDs were fabricated to exhibit a pure blue light emission, peaking at 462 nanometers. The luminance of 691 candela per square meter (the standard unit that specifies brightness of a device) and half-lifetime of 20 minutes achieved in this work are among the best values for pure blue perovskite LEDs based on nanoplatelets reported to date.
“Our work clearly shows the potential of using properly surface passivated perovskite nanoplatelets as emitters for highly efficient and stable LEDs,” Ma said.
Create Blue Light from a Promising Material: Original Article
Efficient and stable emission of warm white light from lead-free halide double perovskites